Low temperature plasma source for medical bioapplications
Technology description
| The name of the technology: | Low temperature plasma source for medical bioapplications |
|---|---|
| Challenge: |
Low temperature plasma (NTP) produces ionized air that has sterilizing effects without having an adverse effect on living tissues. Ozone is also part of the plasma. The main effect of low-temperature plasma is non-specific inhibition of pathogenic microorganisms (bacteria, fungi, yeast) and stimulation of healing. Current NTP sources for medical applications have a number of disadvantages:
|
| Description: |
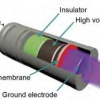
The atmospheric source of low-temperature plasma is intended for use in various medical applications such as disinfection, wound healing, dermatology and dentistry. The application of low-temperature plasma achieves the effect of bacterial inhibition and promotes healing. The new technology shows a unique even distribution of intensity, which also increases the treated area. The shape adapts to the treated area while maintaining an even plasma effect and minimizing risks to the patient. |
| Commercial opportunity: |
Manufacture of medical devices for treatment of chronic wounds (diabetic foot, shin ulcers, pressure ulcers), burns and in other dermatological applications (acne, dermatitis), in surgery or dentistry. The long-term goal of the project presupposes the introduction of a new and promising field of plasma medicine into healthcare in the Czech Republic. Plasma medicine has great potential to significantly influence the possibilities of therapy in a number of medical disciplines and, as a result, to reduce the cost of treatment, improve the prognosis of treatment and thus the quality of life of patients. |
| IP protection status: |
Protected by utility model number: CZ31034 Related patents: CZ304814, CZ306217 |
| Development status: |
Phase 3Corresponds with TRL 5 and TRL 6 Technology validation and implementing it in real environment. Testing the technology outside of the laboratory and its adjustment to external conditions.
|
| Partnering strategy: | Collaboration investment licensing |
| More information: | |
| Images: | |
| Categories: | Medical Devices |
| Institution: | Institute of Experimental Medicine CAS |
| Owner of a technology: |